Predicting and demonstrating of hidden metastable phase in transition metal oxide
Advanced Materials Science and Engineering LEE, JAICHAN Prof. · Bongwook Chung
-

Advanced Materials Science and Engineering LEE, JAICHAN Prof. ·Bongwook Chung
Predicting and demonstrating of hidden metastable phase in transition metal oxide
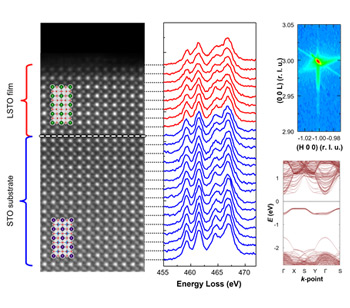
Prof. Jaichan Lee, a professor in the School of Advanced Materials Science and Engineering at Sungkyunkwan University, theoretically predicted the hidden metastable phase in transition metal oxides and experimentally demonstrated it on thin film surfaces. This research predicts and demonstrates the charge-ordered phase, recognized as a fundamental stage for triggering novel functional properties such as superconductivity, colossal magnetoresistance, and multiferroicity. While charge ordering phase transitions have previously been reported in transition metal oxides with highly occupied d orbitals of transition metal cations, this study introduces a significant counterexample. Both theoretical predictions and experimental demonstration confirm the stable presence of charge-ordered phases on the surface of thin films, even within transition metal oxide have low electron occupancy in the d orbitals of transition metal cations. This research holds substantial scientific significance as it reveals that charge ordering can potentially manifest across the complete region of 3d transition metal oxides found on the periodic table. The material studied in this work, strontium titanate (SrTiO3), is a prototypical perovskite transition metal oxide that has been notable for significant novel functional properties, such as the first reported compound oxide superconductor. However, strontium titanate shows relatively weak electron-electron interactions and electrons-lattice coupling, owing to its lowest electron occupancy within the d orbitals of the transition metal titanium. This inherent characteristic of strontium titanate has consequently limited its application as a strongly correlated material for instigating novel functional properties. The researchers initially used the first-principles calculations, to theoretically predict the presence of the charge-ordered metastable phase in lanthanum-doped strontium titanate. Subsequently, they experimentally fabricated lanthanum-doped strontium titanate thin films with a atomically flat surface on strontium titanate (001) substrate, and validiated the stabilization of the metastable charge-ordered phase on the surface of the thin film. The surface triggered stabilization of the charge-ordered phase is both experimentally verified and theoretically predicted by first-principles calculations. Prof. Jaichan Lee explained that the research approach in this study to stabilize metastable phase using a thin film surface is expected to be a fundamental platform to investigate significant novel functional properties induced from charge ordering, such as superconductivity, colossal magnetoresistance, and multiferroicity. This work was supported by the National Research Foundation of Korea and KISTI Supercomputing Center. The research was published in the international multidisciplinary science journal Nature Communications on 8th February. ※Journal: Nature Communications ※Title: Surface triggered stabilization of metastable charge-ordered phase in SrTiO3 ※DOI: 10.1038/s41467-024-45342-8 ※Author list - Corresponding author: Prof. Jaichan Lee, Prof. Chang-Beom Eom - Co-first author: Prof. Kitae Eom, Bongwook Chung - Co-author: Sehoon Oh, Hua Zhou, Jinsol Seo, Sang Ho Oh, Jinhyuk Jang, Si-Young Choi, Minsu Choi, Ilwan Seo, Yun Sang Lee, Youngmin Kim, Hyungwoo Lee, Jung-Woo Lee, Kyoungjun Lee, Mark Rzchowski ▲The verification of the surface triggered charge-ordered phase on the lanthanum-doped strontium titanate thin film surface.
- No. 246
- 2024-04-11
- 231
-

Mechanical Engineering KIM, TAESUNG Prof. ·Hyun Ho Seok
Development of High-Performance Flexible Energy Storage Materials Based on Low-Temperature Plasma
Professor Taesung Kim's research team from the Department of Mechanical Engineering/Department of Nanoscience and Technology/Department of Semiconductor Convergence Engineering, along with Professor Jin Kon Kim's research team at POSTECH and Professor Hong Chul Moon's research team at University of Seoul, have announced the development of a new concept synthesis process technology for producing mesoporous transition metal oxides on flexible substrates by utilizing the synergy effect of low-temperature plasma and thermal processes. Mesoporous metal oxides (MMOs) possess highly advantageous properties such as high surface area and porosity, making them widely used in high-performance energy storage/conversion, sensors, catalysis, etc. However, the conventional synthesis methods require high-temperature sintering processes, making it impossible to synthesize directly on flexible substrates. The widely used soft-template method for MMO fabrication involves the self-assembly of organic support and inorganic precursor to form organic-inorganic composites, followed by the removal of the organic support and the reaction of the inorganic precursor, which traditionally necessitates high-temperature sintering. In this study, the use of low-temperature plasma was employed to lower the synthesis temperature, addressing these issues. The synergy effect between heat and plasma enabled rapid synthesis of various types of MMOs at temperatures of 150-200°C. Additionally, mesoporous vanadium pentoxide (V2O5) was synthesized on a polyimide substrate, realizing flexible energy storage devices. Professor Taesung Kim of Sungkyunkwan University, who developed the low-temperature plasma-based synthesis technology, stated, "The discovery of a method to directly synthesize on flexible substrates in this research will present a new paradigm in the field of smart energy storage/conversion devices." This research was supported by the Ministry of Science, ICT and Future Planning's Creative-Pioneering Researchers Program, Basic Research Program in the field of Science and Technology, and the Nano and Material Technology Development Program (Nano Convergence) and was published in the international journal Advanced Materials on January 19th. ※ Paper Title: Low-Temperature, Universal Synthetic Route for Mesoporous Metal Oxides by Exploiting Synergistic Effect of Thermal Activation and Plasma ※ Authors: Taesung Kim, Jin Kon Kim, Hong Chul Moon (Corresponding author), Geon Woo Kim, Hyun Ho Seok (Co-first authors) ※ DOI: 10.1002/adma.202311809 ■ Development of High-Performance Flexible Energy Storage Materials Based on Low-Temperature Plasma ■ The research was led by Professor Tae Sung Kim from the Department of Mechanical Engineering (corresponding author), and Hyun Ho Seok, a doctoral candidate (first author)
- No. 245
- 2024-04-02
- 411
-

Food Science and Biotechnology WOO, HAN MIN Prof.
Measuring the economic efficiency in Biotechnology
Prof. Woo Han Min, a professor in the Department of Food and Life Science at Sungkyunkwan University, has pioneered the development of an Experiment Price Index (EPI) that quantifies the economic efficiency of the biofoundry technology based on laboratory automation, which is a core manufacturing technique in synthetic biology. This groundbreaking index is the first of its kind globally. The biofoundry technology, akin to custom semiconductor production systems, allows for the design and automation of biological systems. It enables the rapid development of synthetic biology components and cell factories. The Experiment Price Index (EPI) considers research material costs, labor expenses, and the time required for experiments per sample, expressed as the geometric mean. Lower EPI values indicate greater efficiency. Prof. Woo Han Min’s work in quantifying the efficiency of biofoundry through the Experiment Price Index provides a foundation for economically designing and executing large-scale synthetic biology experiments using high-cost automated robots. According to experiments conducted at the Sungkyunkwan University biofoundry Research Center, when comparing 625 synthetic biology experiments (such as gene assembly) performed by human researchers and biofoundry research robots, the latter exhibited approximately twice the efficiency per sample compared to human researchers. Notably, biofoundry research robots reduced experiment time by more than three fold, ultimately demonstrating the capability equivalent to three or more human researchers. This achievement is expected to contribute to the economical establishment of biofoundry infrastructure, especially as biofoundry facilities continue to advance, leading to even higher efficiency Experiment Price Index values. Prof. Woo Han Min envisions that this index will play a crucial role in commercializing synthetic biology-based products by allowing cost-effective planning and execution of large-scale experiments. The research results were published in the online edition of Trends in Biotechnology, a prestigious journal in the field of biotechnology. ※ Journal: Trends in Biotechnology(2024), Impact factor 17.3 (2022), JCR(Journal Citation Reports) Top 1.6% Journals in the Fields of Biotechnology and Applied Microbiolgy ※ Title: Measuring the economic efficiency of laboratory automation in biotechnology ※ DOI: 10.1016/j.tibtech.2024.02.001 ※ 제1저자 및 교신저자: Prof. Woo Han Min(SKKU Synthetic Biology, BioFoundry, Metabolic Engineering) ▲ Measuring the economic efficiency in biotechnology
- No. 244
- 2024-03-27
- 363
-

Integrative Biotechnology BALACHANDRAN, MANAVALAN Prof.
Developed the AI-based tools for identification of virus-induced phosphorylation sites and 2OM sites in human RNA
The CBBL team led by Prof. Balachandran Manavalan at the Department of Integrative Biotechnology, developed two cutting-edge AI-based tools for identification of virus-induced phosphorylation sites and 2OM sites in human RNA. Their innovative approach was published in the top-tier journal Briefings in Bioinformatics (Impact factor 9.5 & JCR = 3). The two methods are as follows: 1. Identification of virus-induced phosphorylation sites using meta-learning approach The global spread of the coronavirus (SARS-CoV-2) has caused significant concern and challenges to global health. Phosphorylation is a common post-translational modification that affects many vital cellular functions and is closely associated with SARS-CoV-2 infection. Accurate identification of phosphorylation sites could provide more in-depth insight into the processes underlying SARS-CoV-2 infection and help alleviate the continuing COVID-19 crisis. However, currently available computational methods in predicting these sites lack accuracy and effectiveness. My team, in collaboration with departmental colleagues including Prof. Young-Jun Jeon, Prof. Minkyung Song, and Prof. Sukchan Lee, developed the novel MeL-STPhos predictor using a meta-learning approach (Figure 1). Specifically, my two PhD students, Nhat Truong Pham and Le Thi Phan created two cell-specific datasets and a generic one using data from Nature and Cell publications. We built a large-scale baseline model (~400) for each dataset, by exploring 29 feature descriptors and 14 different classifiers. The top-performing model from each descriptor was then combined and re-trained for the final prediction. Interestingly, MeL-STPhos generic model has the capability of identifying phosphorylation caused by other viruses, not only SARS-CoV2. Additionally, one cell-specific model accurately detects Threonine phosphorylation sites, showcasing the necessity of multiple models. MeL-STPhos significantly outperformed the best predictor on both datasets, demonstrating the importance of our systematic approach in exploiting different feature descriptors, classifiers, and meta-learning approach, which is responsible for such improved performance. Figure 1. Overview of MeL-STPhos framework. This computational framework includes four steps: Dataset preparation, meta-learning approach, identification of optimal features and classifiers, and web server development. This research was conducted with the support of the Korea Health Technology R&D Project grant through the Korea Health Industry Development Institute (KHIDI) funded by the Ministry of Health & Welfare, Republic of Korea (HI23C0701); National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (2021R1A2C1014338, RS-2023-00217881 and 2021R1C1C1007833) and the result was published in the Briefings in Bioinformatics (https://doi.org/10.1093/bib/bbad433) on December 06, 2023. 2. Identification of 2’-O-methylation sites in human RNA using hybrid deep learning framework 2’-O-methylation (2OM) is the most common post-transcriptional modification of RNA, which plays a crucial role in RNA splicing, RNA stability, and innate immunity. Despite advances in high-throughput detection, the chemical stability of 2OM makes it difficult to detect and map in messenger RNA. While a few bioinformatics tools have made significant advancements in this area, there is still a need for greater accuracy and improvement. My PhD student, Nhat Truong Pham, has developed H2Opred, a novel hybrid learning approach (Figure 2), for accurately identifying 2OM sites. H2Opred incorporated both stacked 1D convolutional neural network (1D-CNN) blocks and stacked attention-based bidirectional gated recurrent unit (Bi-GRU-Att) blocks. 1D-CNN blocks learned effective feature representations from 14 conventional descriptors, while Bi-GRU-Att blocks learned feature representations from five natural language processing-based embeddings extracted from RNA sequences. H2Opred integrated these feature representations to make the final prediction. Moreover, the generic model of H2Opred demonstrated a remarkable performance on both training and testing datasets, significantly outperforming the existing predictor and other four nucleotide specific H2Opred models. Figure 2. Overview of H2Opred framework. This computational framework includes three steps: Dataset preparation, feature extraction and model construction, and feature fusion and web server development. This research was conducted with the support of the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (2021R1A2C1014338 and 2021R1I1A1A01056363), and Korea Health Technology R&D Project grant through the Korea Health Industry Development Institute (KHIDI) funded by the Ministry of Health & Welfare, Republic of Korea (HI23C0701) and the result was published online in the Briefings in Bioinformatics (https://doi.org/10.1093/bib/bbad476) on January 04, 2024. These approaches are not limited in identifying only 2OM or phosphorylation sites. They can also be applied to other research areas, including identification of peptide therapeutic functions and Alzheimer's disease (AD) prediction using gene expression data.
- No. 243
- 2024-03-13
- 3685
-

Medicine LEE, KI YOUNG Prof. ·Jihye Shin
CXCR5 and TLR4 signals synergistically enhance non-small cell lung cancer progression
Lung cancer development and progression are induced by genetic mutations and various factors, such as infections, environment, and lifestyle. Chemokines and their receptors play an important role in the growth and survival of tumor cells, and in cancer development and metastasis. In particular, CXCL13 (Chemokine ligand 13) and its related receptor, CXCR5 (Chemokine receptor 5), are reported to promote metastasis of lung cancer cells through signal transduction pathways, eventually leading to the development and progression of lung cancer. However, the molecular and cellular mechanisms of how CXCR5–CXCL13 functionally regulates lung cancer progression is insufficient. Using data sets of NSCLC patients, we examined whether the expression of CXCR5 is associated with NSCLC progression. In addition, through molecular and cellular mechanism studies, we provided the functional evidence of CXCR5 in NSCLC progression via the CXCR5–CXCL13-TLR4 (Toll-like receptor 4) signaling axis for the activation of NF-κB (Figure 1). Since bacterial or viral infections play a pivotal role for the development and progression of NSCLC, our results strongly demonstrate that the CXCR5–CXCL13-TLR4 signaling axis might be a potential therapeutic target for the intervention of NSCLC patients with up-regulated CXCR5 and TLR4 in precision cancer medicine. Jihye Shin (Sungkyunkwan University College of Medicine, combined master's and doctoral program), Dr. Mijeong Kim (BK21 FOUR young researcher, Sungkyunkwan University College of Medicine), and Jiyoung Kim (combined master's and doctoral program, Sungkyunkwan University College of Medicine) contributed to this study as the first authors. Article: Shin JH, Kim MJ, Kim JY, Kang Y, Kim DH, Jeong SK, Chun E, Lee KY. CXCR5 and TLR4 signals synergistically enhance non-small cell lung cancer progression. Clin Transl Med (IF:10.6). 2024 Jan;14(1):e1547. doi: 10.1002/ctm2.1547. PMID: 38239075. FIGURE 1. CXCR5 and TLR4 signals synergistically enhance lung cancer progression
- No. 242
- 2024-03-04
- 555
-

Biophysics KIM, INKI Prof. ·Dr. Aleksandr Barulin
Biophysics, Prof. Inki Kim Development of metalens system for single molecule detection in real-time
Professor Inki Kim from the Department of Biophysics at Sungkyunkwan University, along with Dr. Aleksandr Barulin, collaborated with Professor Junsuk Rho and doctoral candidates Yeseul Kim and Dong Kyo Oh from the Department of Mechanical Engineering/Chemical Engineering at POSTECH to develop a highly sensitive metalens device capable of real-time observation of single molecule movement at room temperature. Their research was published in the prestigious scientific journal Nature Communications. Single-molecule detection technology is considered a key technology in the fields of biosensing, chemical analysis, molecular dynamics, DNA sequencing, and precision medicine. One of the most commonly used methods among various techniques for detecting single molecules is Fluorescence Correlation Spectroscopy (FCS). FCS analyzes the correlation function of fluorescent signals emitted from molecules to observe the behavior of each molecule individually. In particular, FCS technology is highly sensitive to the characteristics of the lens. Therefore, most reported FCS technologies to date have used high-resolution, high-magnification, ahcromatic, and expensive objective lenses that are properly corrected for aberrations. As the demand for on-site infectious disease diagnosis and personalized/precision medicine increases, scientists are making efforts to develop new biomedical devices such as portable biosensors and miniaturized microscopes that can be integrated with smartphones. However, the lack of technology to miniaturize conventional objective lenses has prevented the development of ultra-compact single-molecule diagnostic devices to date. The research team has developed a highly sensitive metalens device that can observe the movement of single molecules in real-time using a metasurface, which is an ultra-thin flat optical component with a thickness of only 1/1000th of a human hair (Figure 1). For observing single molecules, it is necessary to use a lens with high focusing efficiency and a large numerical aperture, while simultaneously employing aberration corrected high-quality lenses. To meet all these requirements, the research team optimized silicon-based nanostructures and fabricated the device through precise nanofabrication processes. Using the fabricated metalens, the team successfully observed the movement of Alexa 647 single molecules passing through a small space where the focus of light is formed, with a size of 1.6 nm (Figure 2). Furthermore, the research team implemented a technique through FCS analysis to determine the diffusion rate of molecules and the viscosity of the solution. They also developed a technology to distinguish between particles of different sizes, such as quantum dots and nanoparticles (Figure 3). Through this metalens, the possibility of a portable single-molecule detection system was demonstrated for the first time. In future research, the team aims to integrate the metalens into smartphone microscopes and 3D-printed ultra-compact microscopes to realize portable single-molecule measurement systems. Ultimately, it is expected that an on-chip single-molecule detection sensor combining such metalenses and silicon photonics chips could be developed. The research findings were formally published in the prestigious scientific journal Nature Communications (IF = 16.6) on January 2nd. This research was conducted through various funding sources including the K-Brain Project, STEAM: The Future Promising Fusion Technology Pioneer, Engineering Research Center (ERC), Regional Leading Research Center (RLRC), Nanoconnect, POSCO-POSTECH-RIST Convergence Research Center for Flat Optics and Metaphotonics (cFOM), and Sejong Science Fellowship Project. ※ Journal: Dual-wavelength metalens enables epi-fluorescence detection from single molecules ▲ Figure 1. Schematic of the single-molecule detection system based on metalens. ▲ Figure 2. Experimental results of Alexa 647 single molecules measured through metalens fluorescence correlation spectroscopy (FCS). ▲ Figure 3. Various single nano-particle discrimination techniques using metalens FCS.
- No. 241
- 2024-02-16
- 804
-

Food Science and Biotechnology WOO, HAN MIN Prof.
Speeding up the development of microbial cell factories with ‘RNA CRISPR scissors’
Sungkyunkwan University, led by President Yoo Ji-beom, has made a significant stride in biotechnology. Professor Woo Han Min's research team in the Department of Food and Biotechnology has achieved a breakthrough by developing a novel RNA interference system. This cutting-edge system utilizes dead Cas13a RNA CRISPR scissors, previously nonexistent in bacteria. Automated biofoundry technology was employed to enhance the efficiency of developing microbial cell factories. The groundbreaking results of this research were officially published on January 11 through 'Nucleic Acids Research,' a globally recognized academic journal (DOI: 10.1093/nar/gkad1130), with the official announcement made on November 30, 2023. Microbial cell factories, designed for the production of sustainable biofuels, pharmaceuticals, food materials, and chemicals, represent industrial bacteria. These specialized bacteria leverage synthetic biology tools to regulate gene expression and control metabolic processes, aiming to maximize material productivity. This innovative approach holds promise for advancing the production of crucial bio-based products. In details, the study introduces a technology aimed at suppressing the expression of diverse small RNAs within bacterial cells. Leveraging the unique capabilities of dead Cas13a (dCas13a) RNA CRISPR scissors, the researchers successfully developed a method to control both trans-small RNA and cis-small RNA in bacteria. This achievement fills a gap in existing technologies, providing control over small RNAs that were previously challenging to manipulate. Remarkably, this newly developed technology shares similarities with RNA interference mechanisms found in higher organisms, offering the potential to regulate high-level gene expression in bacteria. Moreover, the advanced development of modular loop guide RNA has yielded a technology capable of suppressing target RNA expression across a spectrum ranging from 66% to 92%. This application technology effectively inhibits the expression of polycistronic genes prevalent in bacteria. Unlike existing CRISPRi gene inhibition technology, this approach allows for the targeted suppression of individual polycistronic genes, laying the foundation for the efficient development of cell factories. The RNA CRISPR scissors' bacterial RNA interference technology was applied to create a microbial cell factory dedicated to producing lycopene, known for its antioxidant properties. Biofoundry technology, facilitated by a robot, produced 93 known E. coli sRNAs. A screening process within these libraries identified novel target sRNAs capable of enhancing lycopene productivity. This innovative approach surpasses traditional metabolic engineering, focusing on directly controlling the expression of enzyme genes involved in metabolic reactions. Instead, it introduces a novel metabolic engineering method that regulates downstream gene expression through target sRNA manipulation. Professor Woo Han Min, Director of the Biofoundry Research Center at Sungkyunkwan University, emphasized, "With novel technologies of bacterial RNA interference and biofoundry, we are well-positioned to address diverse challenges using cutting-edge synthetic biology. Our goal is to extend the application of these technologies to new frontiers in medicine, food production, and the manufacturing of high value-added materials. Taking the lead in this endeavor, we are committed to developing advanced cell factories to fulfill these objectives." This research has successfully generated large-scale guide RNA through the integration of bacterial RNA interference technology and biofoundry technology—a pivotal component in biomanufacturing. This transformative combination allows for the reprogramming of bacteria to serve as efficient cell factories, facilitating the screening of target materials and automating the entire Design-Build-Test-Learn (DBTL) cycle, a fundamental philosophy in synthetic biology research. Consequently, the development of cell factories is expedited, marking a significant advancement in the field. Meanwhile, the research results were published online through the renowned academic journal 'Nucleic Acids Research' on November 28th. This research achievement was supported by the National Research Foundation of Korea’s Senior Researchers, Basic Research Laboratory Support Program, Microbial Control and Application Core Technology Development Project, and the Ministry of Science and ICT-supported Petrochemical Alternative Eco-friendly Chemical Technology Development Project, aimed at leading the biochemistry industry through the development of next-generation biorefinery core technologies. The underlying technology was domestically patented, and completed in 2022 (patent registration number 10-2422842). ※ Journal: Nucleic Acids Research (2023), Impact factor 14.9 (2022), Ranked in the top 3.3% in the field of JCR Biochemistry and Molecular Biology ※ Title: CRISPR-dCas13a system for programmable small RNAs and polycistronic mRNA repression in bacteria ※ DOI: 10.1093/nar/gkad1130 ※ First Author: Ph.D. Ko Sung-chun (SKKU, Department of Food and Biotechnology) ※ Corresponding Author: . Professor Woo Han Min(SKKU, Department of Food and Biotechnology, the Biofoundry Research Center, Metabolic Engineering) ▲RNA CRISPR scissors' bacterial RNA interference technology was applied to create a microbial cell factory
- No. 240
- 2024-02-13
- 714
-

SKKU Advanced Institute of Nano Technology LEE, JIN WOOK Prof. ·Keonwoo Park
Atmospheric Humidity Underlies Irreproducibility of Formamidinium Lead Iodide Perovskites
The research team led by Prof. Jin-Wook Lee in the SKKU Advanced Nano Technology (SAINT), and Department of Nano Engineering has highlighted the significant influence of atmospheric humidity on the reproducibility and quality of perovskite solar cells (PSCs) during fabrication processes. Their related research paper was published online on December 21st in the international scientific journal 'Advanced Materials.' Perovskite solar cells, known for their high energy conversion efficiency of over 26.1% and the ability to be produced through cost-effective solution processes, have garnered significant attention as next-generation solar cells for the upcoming era of carbon neutrality. While research efforts to accelerate their commercialization are underway globally, research laboratories, startups, and large corporations often face challenges due to unstable fabrication environments, resulting in poor reproducibility of perovskite film quality. This inconsistency in film quality is considered a major obstacle to commercialization, leading to skepticism in data interpretation. In the process of fabricating the world's high-record perovskite film, formamidinium lead triiodide (FAPbI3) perovskite is used as an essential material, and the highly volatile Methylammonium chloride (MACl) has been treated as indispensable additive for enhancing the stability and crystallinity of FAPbI3. Prof. Jin-Wook Lee's research team considered the high volatility and hygroscopic nature of MACl, discovering that atmospheric humidity plays a crucial role in improving the optoelectronic properties and quality of perovskite films. Specifically, the spin-coating process in a dry environment (under an N2 atmosphere) beneficially induces well stabilized nucleation and crystallization of perovskite within the MACl additive, forming highly crystalline perovskite films. However, it was observed that MACl is not effectively removed during the annealing process, leading to the formation of interstitial defects within the perovskite lattice, causing phase instability and a reduction in device efficiency. The research team revealed that exposure of the perovskite film to moderate atmospheric humidity during the annealing process can induce an azeotropic effect that lowers the vaporization point of MACl, aiding in its facilitative removal. This, in turn, contributes to the successful improvement of device conversion efficiency and phase stability. Furthermore, Prof. Jin-Wook Lee's research team has presented an intriguing perspective that, contrary to the conventional belief that atmospheric humidity is a sensitive and harmful factor to be avoided in the perovskite fabrication process, it can actually be beneficial for the perovskite manufacturing process. This perspective could potentially enhance scalability, reduce production costs, and improve accessibility, all of which are crucial factors for commercialization. Additionally, Prof. Jin-Wook Lee’s research team emphasizes the importance of specifying the process atmosphere for successful and rapid commercialization. ※ Related research papers and website 1) Atmospheric Humidity Underlies Irreproducibility of Formamidinium Lead Iodide Perovskites, Advanced Material, 2023, https://doi.org/10.1002/adma.202307265 (corresponding author: Prof. Jin-Wook Lee, first-author: Keonwoo Park) 2) Prof. Jin-Wook Lee`s Lab website: https://jwlee870217.wixsite.com/mysite Figure 1. Atmospheric humidity affects the spin-coating and annealing processes, demonstrating its influence on nucleation and the efficiency of the resulting devices.
- No. 239
- 2024-01-24
- 591
-

School of Medicine/Department of Metabiohealth LEE, SEONGHYUN Prof.
Engineered mitochondrial adenine base editor with reduced off-target effects on RNA with an animal model for genetic dis
Professor Seonghyun Lee (Department of Precision Medicine, School of Medicine/Department of Metabiohealth) successfully developed engineered mitochondrial base editor and created mice with adenine base modifications at specific sequences of mitochondrial DNA, marking the world's first achievement. This research was conducted through a collaborative effort involving Yonsei University College of Medicine, KIST Brain Science Institute, Korea University College of Medicine, and Edgene Incorporation. The mitochondria, existing as the energy source within cells, possess mitochondrial DNA that carries the genetic information for proteins essential in energy metabolism within its structure. Defects in this DNA lead to mitochondrial dysfunction, manifesting as various disorders in the brain, nerves, and muscles. Additionally, due to the unique maternal inheritance of mitochondria, maternal mitochondrial defects can be passed down to offspring, resulting in mitochondrial diseases in the descendants. Current CRISPR-Cas9 gene editing technology is widely used for DNA correction, but it has limitations in mitochondrial DNA correction due to the inability of the guide RNA, which is used to recognize specific DNA sequences, to be transported into the mitochondria. As of now, the developed mitochondrial DNA correction technologies include DdCBE (Nature, 2020), capable of correcting C to T among the four DNA base sequences, and TALED (Cell, 2022), capable of correcting A to G. While there are reported cases of creating mice with mitochondrial C-to-T gene correction using DdCBE, there have been no reported instances of successful A-to-G gene correction in mitochondrial DNA in animal experiments. The research team confirmed that the previously developed mitochondrial DNA editing technology, TALED, induces unintended random RNA mutations within cells. They discovered that when TALED is injected into mouse oocytes, normal embryo development does not occur. Therefore, they improved the TALED by engineering protein for precise DNA modification, resulting in the development of V28R-TALED. Through this improvement, the team observed a significant reduction in the unintended RNA mutations within cells, which were side effects of TALED. Furthermore, by microinjecting the improved TALED into mouse oocytes, the researchers successfully created mice carrying mutations associated with Leigh syndrome, a mitochondrial disorder, and exhibiting symptomatic features of the disease. This study was published in the prestigious international journal, Cell (IF=66.85), on January 4, 2024 (Korean Standard Time). Title: Engineering TALE-linked deaminases to facilitate precision adenine base editing in mitochondrial DNA DOI: TBA Author: Prof. Seonghyun Lee (Corresponding Author, Assistant Professor in Department of Precision Medicine, School of Medicine & Department of Metabiohealth) Figure Schematic Drawing of Engineered TALED and its application
- No. 238
- 2024-01-16
- 957
-

Advanced Materials Science and Engineering KIM, HANKI Prof. ·Hae-Joon Seok
Professor Kim Han-Ki's research group has developed specialized low-resistance/high-transparency ITO transparent electro
Sungkyunkwan University announced that Professor Kim Han-Ki's research group in the Department of Advanced Materials Engineering developed specialized ITO transparent electrode technology for the commercialization of next-generation large-area high-efficiency perovskite solar cells. ※Journal: Advanced Energy Materials (Impact factor: 29.698) Title: Sn composition engineering toward the breakthrough of transparent front electrodes for efficient and stable perovskite solar cells. ※Author names: Han-Ki Kim(Corresponding author), Hae-Joon Seok (1st author) Overcoming the Problems and Limitations of Previous Research: The existing research on perovskite solar cells faced limitations in achieving maximum efficiency because it relied on commercial ITO transparent electrodes doped with 10 wt.% of Sn, commonly used in displays or basic solar cells, without dedicated transparent electrodes. Research on the Sn dopant content, taking into account the characteristics of the perovskite solar cell's active layer and buffer layer, had not been conducted. Consequently, most studies utilized ITO electrodes intended for display purposes. Professor Kim Han-Ki's research group addressed this by implementing co-sputtering processes to finely control the Sn dopant content and introducing rapid thermal annealing using light to achieve the world's first dedicated ITO transparent electrode specialized for perovskite solar cells. Named CE-ITO (Composition Engineered ITO), this perovskite solar cell-dedicated transparent electrode exhibited a low sheet resistance of 2.75 Ohm/square and 94% optical transmittance. The developed technology allows for a significant enhancement in perovskite solar cell efficiency, increasing it from 20.78% (with a typical ITO electrode) to 23.35% (with CE-ITO electrode). Additionally, perovskite solar cells manufactured with CE-ITO electrodes demonstrated superior stability compared to those produced with conventional ITO, securing a key technology for the commercialization of next-generation perovskite solar cells. While most research teams focused on improving efficiency through studies on the active layer or buffer layer of perovskite solar cells, Professor Kim Han-Ki's group took a novel approach by significantly enhancing the characteristics of the transparent electrode. The outstanding electrical/optical properties and stability of the CE-ITO electrode developed by Professor Kim Han-Ki's research group are attributed to precise control of the Sn dopant content, considering the energy band of the perovskite active layer, and crystallization technology facilitated by rapid thermal annealing. The team explains that this not only enhances the interfacial properties of the perovskite solar cell's layered structure but also improves the crystallinity of the layer deposited on top of it, demonstrating an overall improvement in the device's performance. These research results are expected to advance the research and commercialization of perovskite solar cells by providing crucial electrode technology. It is anticipated that this technology will accelerate the commercialization of semi-transparent perovskite solar cells and ultra-efficient tandem solar cells. Moreover, the application of CE-ITO technology is expected to be viable for ensuring the stability of next-generation high-efficiency solar cells for space applications. Author's Comment: Through a paradigm shift, our research team has developed a technology that significantly enhances the characteristics of ITO transparent electrodes, which is not commonly studied, unlike the research on the active layer or buffer layer materials. This innovation aims to maximize the efficiency of next-generation perovskite solar cells. The CE-ITO technology developed by our research team is not only applicable to perovskite solar cells but can also be applied to inorganic displays, OLEDs, smart windows, touch panels, biosensors, and transparent electronic components. Therefore, we anticipate that this core technology can dramatically improve the characteristics of next-generation optoelectronic devices through the replacement of electrodes. The CE-ITO technology is being prepared for mass production as a test element group (TEG) product for research purposes through Professor Kim Han-Ki's laboratory startup company, Coconutmaterials (http://www.coconutmaterials.com). It is expected to be supplied to universities, research institutes, and companies engaged in perovskite solar cell research. Research support and Publication: This research was conducted with the support of the Ministry of Science and ICT's Research Material Development Diffusion Project and the Gyeonggi Regional Research Center (GRRC). This research was published in the international scientific journal Advanced Energy Materials (IF: 29.698) on December 10th. Flexible CEITO thin film deposited on UTG (Ultra Thin Glass) Comparison of the interface of perovskite solar cells fabricated on conventional ITO and CEITO Adv. Energy. Mater. (2023)
- No. 237
- 2024-01-05
- 1032
-

Biological Sciences LEE, SANGHO Prof. ·Mr. Yeongmok Lee
Deciphering the secret of drought tolerance, unveiled activation mechanism of plant anion channel
The research team led by Professor Sangho Lee in the department of biological sciences (first author Yeongmok Lee) has uncovered a crucial puzzle piece regarding how plants respond to environmental stress and survive through the collaboration with Korea Brain Research Institute, the Korea Basic Science Institute and the Rural Development Administration. They elucidated the structural basis for the activation mechanism of the SLAC1 anion channel, a master switch of plant stomatal regulation, using cryo-electron microscopy (cryo-EM). Tiny pores called stomata exist on the surface of plant leaves, allowing the absorption of carbon dioxide for photosynthesis and the release of oxygen. However, stomata can cause significant water loss during the drought and serve as entry points for pathogens. To overcome the limitations, plants have evolved the SLAC1 anion channel, which regulate stomatal closure in response to drought, pathogens, carbon dioxide, ozone, and other environmental factors. However, reports indicate that the precise stomatal regulation is being disrupted due to rapid increases in temperature and carbon dioxide resulting from climate change. Given the importance of stomatal regulation, extensive researches have been conducted over the past decade to understand how the SLAC1 responds to stress and becomes activated. However, activation mechanism of the SLAC1 remains elusive and two alternative hypotheses have been suggested from previous studies. The research team determined the SLAC1 structures in open and closed states using cryo-EM and proposed a novel activation mechanism that accommodates two hypotheses from previous studies. This study revealed a two-step activation process involving phosphorylation-induced inhibition release and subsequent binding activation. Prof. Sangho Lee remarked, "Through this study, the mystery of how plants detect stress and regulate stomata has finally been unveiled. This knowledge opens up the possibility for various applied researches, such as climate change adaptation and stress resistance through stomatal regulation." He also said, “It shows a research case using cryo-EM, a state-of-the-art device recently introduced in our university, emphasizing its potential for producing outstanding research outcomes.” The research was published online in Nature Communications (IF: 17.694) on November 14th. ※ Title: Cryo-EM structures of the plant anion channel SLAC1 from Arabidopsis thaliana suggest a combined activation model ※ DOI: https://doi.org/10.1038/s41467-023-43193-3 ※ Author: Yeongmok Lee (first author), Seoyeon Jung, Chi Truc Han Le, Prof. Sangho Lee (corresponding author) Figure 1. Open and closed structures of anion channel SLAC1 Figure 2. A combined activation mechanism of anion channel SLAC1
- No. 236
- 2023-12-26
- 1396
-

SKKU Advanced Institute of Nano Technology LIM, YONGTAIK Prof. ·Shin Hong sik
Development of technology to overcome immune tolerance of small molecular immunotherapy drugs
Sungkyun Institute for Nanoscience and Technology (SAINT) Professor Lim Yong taik's research team (first author Shin Hong sik, a doctoral student) has developed a Toll-like receptor 7/8 agonist (TLR 7/8a) adjuvant that can convert the immunosuppressive environment of the tumor microenvironment into an immune-activating environment with minimal toxicity. toll-like receptor agonist adjuvant was developed. In particular, the immune tolerance phenomenon associated with TLR 7/8a was overcome through molecular and macroscale coordination of the drug delivery system, thereby exceeding the limitations of existing Toll-like receptor 7/8 agonists.The research group also used the principles to overcome the limitations of current macrophage-based cell therapy by conjugating TLR 7/8a onto macrophage cells through click chemistry. TLR 7/8a have received attention for their ability to modulate not only innate immune activation but also the immunosuppressive environment of the tumor microenvironment, but they face great difficulties in actual clinical application due to their unique systemic toxicity and immune tolerance. . To overcome these limitations, Professor Lim Yong taik’s research team developed Nanoliposome(pro-TLR7/8a) (NL(pro-TLR7/8a)), a new concept adjuvant material that can overcome both systemic toxicity and immune tolerance phenomenon through molecular-scopic and macroscopic coordination of drug delivery systems. NL (pro-TLR7/8a) molecular-scopically masks the point of action of the Toll-like receptor 7/8 agonist molecule with a cholesterol molecule, thereby minimizing the toxicity problem of non-specific action, and in a specific environment, the masked cholesterol gradually recovered to a toll-like receptor 7/8 agonist. It is a drug delivery vehicle that can gradually recover the original activity of the receptor 7/8 agonist and overcome the immune tolerance response caused by an excessive immune response. NL(pro-TLR7/8a) modulates the immunosuppressive environment of the tumor microenvironment into an immune active environment, promoting continuous secretion of immune active cytokines (interleukin 12, interferon gamma) and minimized immune active cytokines (interleukin 6) in the blood, which are indicators of systemic toxicity. NL(pro-TLR7/8a) showed excellent therapeutic effects in several tumor models (skin cancer, lung cancer, breast cancer) and achieved complete tumor regression in combination treatment with the immune checkpoint inhibitors anti-PD-1 and anti-CTLA-4. Combination therapy with Doxorubicin, an anticancer drug currently applied clinically, also showed excellent therapeutic effects, showing high potential for clinical application. Article Name : Molecular Masking of Synthetic Immunomodulator Evokes Antitumor Immunity with Reduced Immune Tolerance and Systemic Toxicity by Temporal Recovery of Activity and Sustained Stimulation (Advanced Material (IF=32.086), Oct 30, 2023) Author : Hong sik Shin (1st author, Ms, Ph.D integrated student), Sohyun Kim (Co-1st author, Ph.D), Seung Mo Jin (Co-author, Ph.D student), Yeon Jeong Yoo (Co-author, Ph.D student), Janghun Heo (Co-author, Ms, Ph.D integrated student) and Yong Taik Lim (Corresponding author, Sungkyunkwan Uni. Prof.) Article Name : Nanoengineered Macrophages Armed with TLR7/8 Agonist Enhance Remodeling of Immunosuppressive Tumor Microenvironment (Small (IF : 15.153), Nov 15, 2023) Author : Yeon Jeong Yoo (1st author, Ph.D student), Suhyun Kim (Co-author, Ms student), Sei Hyun Park (Co-author, Ph.D student), Janghun Heo (Co-author, Ms, Ph.D integrated student) and Yong Taik Lim (Corresponding author, Sungkyunkwan Uni. Prof.)
- No. 235
- 2023-12-06
- 1720

